
Hydrogen-3, symbolized 3H, is also called tritium and sometimes symbolized T. Hydrogen-2, symbolized 2H, is also called deuterium and sometimes symbolized D. Note that in addition to standard names and symbols, the isotopes of hydrogen are often referred to using common names and accompanying symbols. Information about the naturally occurring isotopes of elements with atomic numbers 1 through 10 is given in. These atoms are called radioactive isotopes or radioisotopes. The new atoms created may be in a high energy state and emit gamma rays which lowers the energy but alone does not change the atom into another isotope. Neutrons, protons, and positrons can also be emitted and electrons can be captured to attain a more stable atomic configuration (lower level of potential energy ) through a process called radioactive decay. Unstable isotopes most commonly emit alpha particles (He 2+) and electrons. Carbon-14 ( 14C) is unstable and only occurs in trace amounts. Carbon-12 ( 12C) is the most abundant of the carbon isotopes, accounting for 98.89% of carbon on Earth. While the mass of individual isotopes is different, their physical and chemical properties remain mostly unchanged. It is the second most abundant element in the Earths crust, making up 28 of it by. The symbol for an atom indicates the element via its usual two-letter symbol, the mass number as a left superscript, the atomic number as a left subscript (sometimes omitted), and the charge as a right superscript. Silicon is a metalloid element belonging to group 14 of the periodic table. The first alloy used on a large scale was bronze, made of tin and copper, from as early as 3000 BC. It is obtained chiefly from the mineral cassiterite, which contains tin dioxide. They differ only because a 24Mg atom has 12 neutrons in its nucleus, a 25Mg atom has 13 neutrons, and a 26Mg has 14 neutrons. Tin is a post-transition metal in group 14 of the periodic table. 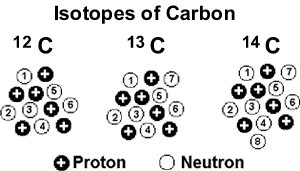
Note that isotope symbols are read as “element, mass number.” For instance, in the case of magnesium, 24Mg is read as “magnesium 24,” and can be written as “magnesium-24” or “Mg-24.” All magnesium atoms have 12 protons in their nucleus. Carbon-14 (or 14C) contains six protons, eight neutrons, and six electrons its atomic mass is 14 amu (six protons and eight neutrons).Carbon-12 (or 12C) contains six protons, six neutrons, and six electrons therefore, it has a mass number of 12 amu (six protons and six neutrons). 
The atomic number is sometimes written as a subscript preceding the symbol, but since this number defines the element’s identity, as does its symbol, it is often omitted. The symbol for a specific isotope of any element is written by placing the mass number as a superscript to the left of the element symbol, as shown in the figure below. Isotopes are defined first by their element and then by the sum of the protons and neutrons present. Some elements, such as carbon, potassium, and uranium, have multiple naturally-occurring isotopes. Isotopes are various forms of an element that have the same number of protons but a different number of neutrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
